MATTER
Chemists study the structures,
physical properties, and chemical properties of material substances. These
consist of matter, which is
anything that occupies space and has mass. Gold and iridium are matter, as are
peanuts, people, and postage stamps. Smoke, smog, and laughing gas are matter.
Energy, light, and sound, however, are not matter; ideas and emotions are also
not matter.
The mass of
an object is the quantity of matter it contains. Do not confuse an object’s
mass with its weight, which is a
force caused by the gravitational attraction that operates on the object. Mass
is a fundamental property of an object that does not depend on its location.In
physical terms, the mass of an object is directly proportional to the force
required to change its speed or direction. A more detailed discussion of the
differences between weight and mass and the units used to measure them is
included in Essential Skills 1 (Section 1.9). Weight,
on the other hand, depends on the location of an object. An astronaut whose
mass is 95 kg weighs about 210 lb on Earth but only about 35 lb on the moon
because the gravitational force he or she experiences on the moon is
approximately one-sixth the force experienced on Earth. For practical purposes,
weight and mass are often used interchangeably in laboratories. Because the
force of gravity is considered to be the same everywhere on Earth’s surface,
2.2 lb (a weight) equals 1.0 kg (a mass), regardless of the location of the
laboratory on Earth.
Under normal conditions, there are
three distinct states of matter: solids, liquids, and gases. Solids are relatively rigid and have
fixed shapes and volumes. A rock, for example, is a solid. In contrast, liquids have fixed volumes but flow to
assume the shape of their containers, such as a beverage in a can. Gases, such as air in an automobile tire,
have neither fixed shapes nor fixed volumes and expand to completely fill their
containers. Whereas the volume of gases strongly depends on their temperature
and pressure (the amount of
force exerted on a given area), the volumes of liquids and solids are virtually
independent of temperature and pressure. Matter can often change from one
physical state to another in a process called a physical change. For example, liquid water
can be heated to form a gas called steam, or steam can be cooled to form liquid
water. However, such changes of state do not affect the chemical composition of
the substance.
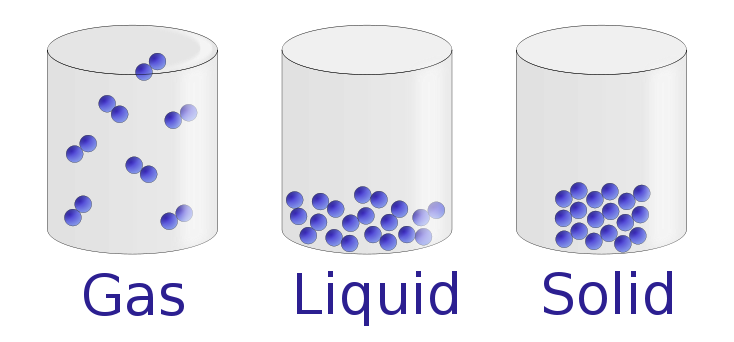
Figure \(\PageIndex{1}\): The Three States of Matter. Solids have a defined
shape and volume. Liquids have a fixed volume but flow to assume the shape of
their containers. Gases completely fill their containers, regardless of volume.
Figure used with permission from Wikipedia
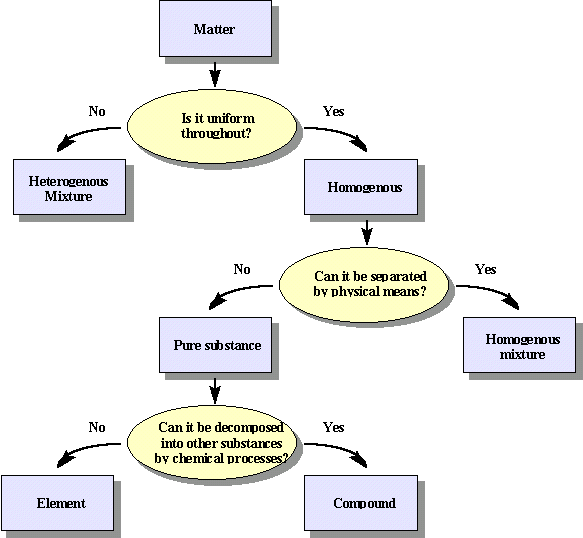
Pure Substances and Mixtures
A pure
chemical substance is any matter that has a fixed chemical composition and
characteristic properties. Oxygen, for example, is a pure chemical substance
that is a colorless, odorless gas at 25°C. Very few samples of matter consist
of pure substances; instead, most are mixtures, which are combinations of
two or more pure substances in variable proportions in which the individual
substances retain their identity. Air, tap water, milk, blue cheese, bread, and
dirt are all mixtures. If all portions of a material are in the same state,
have no visible boundaries, and are uniform throughout, then the material
is homogeneous. Examples of homogeneous mixtures are the air we
breathe and the tap water we drink. Homogeneous mixtures are also
called solutions. Thus air is a solution of nitrogen, oxygen, water vapor,
carbon dioxide, and several other gases; tap water is a solution of small
amounts of several substances in water. The specific compositions of both of
these solutions are not fixed, however, but depend on both source and location;
for example, the composition of tap water in Boise, Idaho, is not the
same as the composition of tap water in Buffalo, New York. Although most
solutions we encounter are liquid, solutions can also be solid. The gray substance
still used by some dentists to fill tooth cavities is a complex solid solution
that contains 50% mercury and 50% of a powder that contains mostly silver, tin,
and copper, with small amounts of zinc and mercury. Solid solutions of two or
more metals are commonly called alloys.
If
the composition of a material is not completely uniform, then it is heterogeneous (e.g.,
chocolate chip cookie dough, blue cheese, and dirt). Mixtures that appear to be
homogeneous are often found to be heterogeneous after microscopic examination.
Milk, for example, appears to be homogeneous, but when examined under a
microscope, it clearly consists of tiny globules of fat and protein dispersed
in water. The components of heterogeneous mixtures can usually be separated by
simple means. Solid-liquid mixtures such as sand in water or tea leaves in tea
are readily separated by filtration, which consists of passing the mixture
through a barrier, such as a strainer, with holes or pores that are smaller
than the solid particles. In principle, mixtures of two or more solids, such as
sugar and salt, can be separated by microscopic inspection and sorting. More
complex operations are usually necessary, though, such as when separating gold
nuggets from river gravel by panning. First solid material is filtered from
river water; then the solids are separated by inspection. If gold is embedded
in rock, it may have to be isolated using chemical methods.

Figure \(\PageIndex{2}\): A Heterogeneous Mixture. Under a microscope,
whole milk is actually a heterogeneous mixture composed of globules of fat and
protein dispersed in water. Figure used with permission from Wikipedia
Homogeneous mixtures (solutions) can
be separated into their component substances by physical processes that rely on
differences in some physical property, such as differences in their boiling
points. Two of these separation methods are distillation and
crystallization. Distillation makes
use of differences in volatility, a measure of how easily a substance is
converted to a gas at a given temperature. A simple distillation apparatus for
separating a mixture of substances, at least one of which is a liquid. The most
volatile component boils first and is condensed back to a liquid in the
water-cooled condenser, from which it flows into the receiving flask. If a
solution of salt and water is distilled, for example, the more volatile
component, pure water, collects in the receiving flask, while the salt remains
in the distillation flask.

Figure \(\PageIndex{3}\): The Distillation of a Solution of Table Salt
in Water. The solution of salt in water is heated in the distilling flask until
it boils. The resulting vapor is enriched in the more volatile component
(water), which condenses to a liquid in the cold condenser and is then
collected in the receiving flask.
Mixtures of
two or more liquids with different boiling points can be separated with a more
complex distillation apparatus. One example is the refining of crude petroleum
into a range of useful products: aviation fuel, gasoline, kerosene, diesel
fuel, and lubricating oil (in the approximate order of decreasing volatility).
Another example is the distillation of alcoholic spirits such as brandy or
whiskey. This relatively simple procedure caused more than a few headaches for
federal authorities in the 1920s during the era of Prohibition, when illegal
stills proliferated in remote regions of the United States.
Crystallization separates mixtures based on
differences in solubility, a measure of how much solid substance remains
dissolved in a given amount of a specified liquid. Most substances are more
soluble at higher temperatures, so a mixture of two or more substances can be
dissolved at an elevated temperature and then allowed to cool slowly.
Alternatively, the liquid, called the solvent, may be allowed to
evaporate. In either case, the least soluble of the dissolved substances, the
one that is least likely to remain in solution, usually forms crystals first,
and these crystals can be removed from the remaining solution by filtration.

Figure
\(\PageIndex{4}\): The Crystallization of
Sodium Acetate from a Concentrated Solution of Sodium Acetate in Water. The
addition of a small “seed” crystal (a) causes the compound to form white
crystals, which grow and eventually occupy most of the flask.
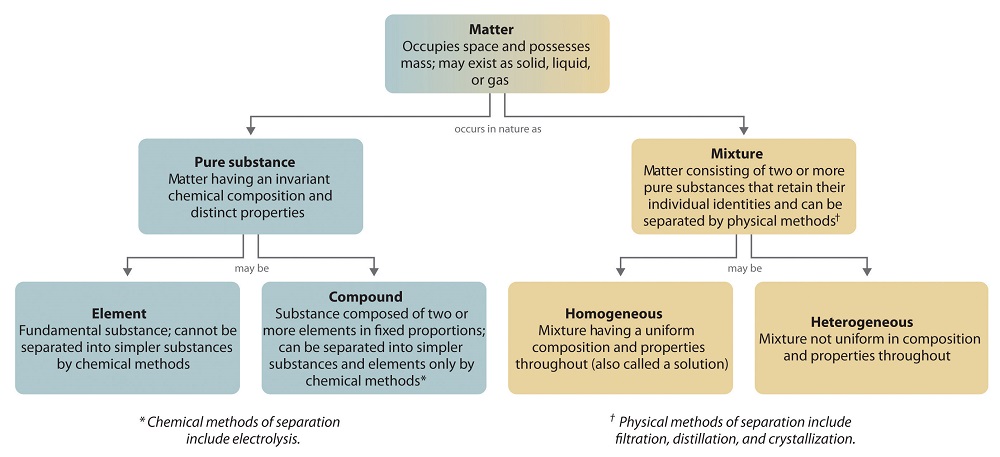
Most
mixtures can be separated into pure substances, which may be either elements or
compounds. An element, such as gray,
metallic sodium, is a substance that cannot be broken down into simpler ones by
chemical changes; a compound,
such as white, crystalline sodium chloride, contains two or more elements and
has chemical and physical properties that are usually different from those of
the elements of which it is composed. With only a few exceptions, a particular
compound has the same elemental composition (the same elements in the same
proportions) regardless of its source or history. The chemical composition of a
substance is altered in a process called a chemical
change. The conversion of two or more elements, such as sodium and
chlorine, to a chemical compound, sodium chloride, is an example of a chemical
change, often called a chemical reaction. Currently, about 115 elements are
known, but millions of chemical compounds have been prepared from these 115
elements. The known elements are listed in the periodic table.
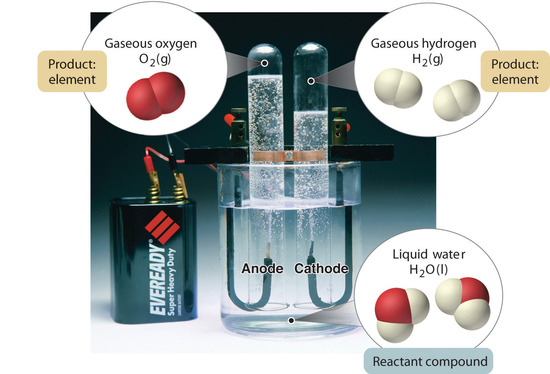
Figure
\(\PageIndex{5}\): The Decomposition of Water
to Hydrogen and Oxygen by Electrolysis. Water is a chemical compound; hydrogen
and oxygen are elements.
In general,
a reverse chemical process breaks down compounds into their elements. For
example, water (a compound) can be decomposed into hydrogen and oxygen (both
elements) by a process calledelectrolysis. In electrolysis, electricity
provides the energy needed to separate a compound into its constituent elements
(Figure \(\PageIndex{5}\)). A similar technique is
used on a vast scale to obtain pure aluminum, an element, from its ores, which
are mixtures of compounds. Because a great deal of energy is required for
electrolysis, the cost of electricity is by far the greatest expense incurred
in manufacturing pure aluminum. Thus recycling aluminum is both cost-effective
and ecologically sound. The overall organization of matter and the methods used
to separate mixtures are summarized in Figure
\(\PageIndex{6}\).
so what difference heterogeneous and homogeneous mixture? give me examples.
BalasHapusHetrogeneous mixtures are mixtures are those in which the components are not evenly distributed over the major component/constitute of the mixture
HapusThe very (unevenly distributed) form of heterogeneous mixtures is the reason why most people shake the mixtures before using them
Ex. Milk of magnesia, copper sulphate
Homogeneous mixuters are those in which the components are evenly distrubuted over the major component/constitute of the mixture
Ex. Blood, milk
Most mixtures can be separated into pure substances, which may be either elements or compounds in the periodic table. how to separate into pure substances?
BalasHapusPure substances are substances that have a fixed and uniform chemical composition throughout its parts such as water, air, nitrogen and carbon dioxide. Substances that do not have a uniform chemical composition such as in oil and water mixtures are not pure substances.
HapusSubstances can have several types of phases such as solids, liquids or gases depending on the conditions of the substance. For example, at room temperature and pressure, water will be in the liquid phase, nitrogen in the gas phase while aluminum is in a solid phase.
There is, however, a situation in which two phases of pure substance occur in equilibrium. For example, water is a mixture of liquid and vapor phase in the boiler and condenser in a steam generator. Similarly, the Refrigerant changes from a liquid to a gas in a refrigerator freezer.
In our discussion, water will be used as an example to illustrate the basic principles that occur in pure substances under various conditions along with the properties of the substances attached to them
"Very few samples of matter consist of pure substances" so what the different between matter and pure subtances?
BalasHapusMatter is everything around you. Atoms and molecules are all composed of matter. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. We talk about the difference between mass and weight in another section. Matter is sometimes related to light and electromagnetic radiation.
HapusEven though matter can be found all over the Universe, you will only find it in a few forms on Earth. We cover five states of matter on the site. Each of those states is sometimes called a phase. There are many other states of matter that exist in extreme environments. Scientists will probably discover more states as we continue to explore the Universe.
Pure Substance Definition
A pure substance is a sample of matter with both definite and constant composition with distinct chemical properties.
Alternate Spellings: chemical substance
Examples of Pure Substances
Examples of pure substances include elements and compounds. Alloys and other solutions may also be considered pure.
diamond
gold
table salt (sodium chloride)
ethanol
brass
bronze
saline solution
please explain again of the example heterogenous and homogenous coumpounds??
BalasHapusThe homogeneous mixture is a mixture in which there are compositions and properties of the same substance. Examples such as air, air is a collection of substances that have homogeneous gas properties. One tablespoon of salt is put into a glass of water. Which is then stirred until evenly distributed, this can also be called a homogeneous mixture.
HapusHeterogeneous mixture is a mixture consisting of two or more substances having different properties. Examples such as those mentioned are sand and water. This mixture is an example of a heterogeneous mixture, the reason being that it consists of a collection of substances of a different nature, the sand has a solid nature whereas water has a liquid nature.
Heterogeneous mixture is divided into 2, namely suspension and colloid. Suspension is a mixture of substances or compounds that look cloudy and unstable, if left unchecked. While the colloid is a mixture of substances that can not be mixed evenly and if left unchecked
I would like to ask, on what terms can be a compound of an element?
BalasHapusPlease explain.
Compounds are substances formed from the incorporation of elements with certain divisions. Compounds are produced from chemical reactions between two or more elements through forming reactions. For example, iron rust (hematite) of Fe2O3 is produced by the reaction of iron (Fe) with oxygen (O). The compounds can be broken down into their constituent elements by the decomposition reaction.
HapusCompounds have properties different from the constituent elements. Compounds can only be decomposed into their constituent elements by chemical reactions. Under the same conditions, the compound may have a different form with the constituent elements. The physical and chemical properties of the compounds are different from the constituent elements. For example the reaction between hydrogen gas and oxygen gas to form water compounds in the form of liquid.
Characteristics of the Compound:
Is a single substance
Formed from two or more elements of a different type with a certain and fixed ratio
The nature of the compound is different from the properties of its constituent elements
The compound can be decomposed into its elements by chemical means
Properties of the Compound:
A. The compound may be formed by a process of a chemical reaction
B. The constituent components present in a compound must have a certain ratio of a fixed nature.
C. The compound will not be separated by its constituent components back by a physical reaction.
D. The compound can be categorized as a single substance compound.
E. Has certain properties that are different from the constituent elements. Comparison of two hydrogens and one oxygen.
you try to explain again and give examples of what is Pure Substances and Mixtures?
BalasHapusPure substances are substances that have a constant (homogeneous) composition and are of the same (consistent) properties throughout their parts. Examples of pure substances are tin, sulfur, diamond, water, pure sugar (sucrose), salts Table (sodium chloride) and baking soda (sodium bicarbonate). This pure substance can be an element or a compound.
HapusTin, sulfur and diamond are examples of pure substances in the form of chemical elements. All elements are pure substances. Water, sugar, salt and baking soda are pure substances that are chemical compounds. Chemical compounds are also pure substances.
A substance that is something that occupies space and has mass.
Solid substances:
1. iron
2. Nickel
3. Aluminum
4. gold
5. silver
Liquid:
1. water
2. alcohol
3. mercury
4. syrup
5. soy sauce
Gas substance:
1. oxygen
2. Nitrogen
3. helium
4. ozone
5. fluorescence
Explain the difference between a mixture of homogeneous and heterogeneous mixtures?
BalasHapus
Hapus1.) Heterogeneous Definition: A heterogeneous mixtures is one which has a non-uniform composition. Examples: A mixture of sand and water is heterogenous. Concrete is heterogeneous.
2.) Homogeneous Definition: Homogeneous refers to a substance that is consistent or uniform throughout its volume. Examples: Air is considered a homogeneous mixture of gases.
could you give an example of mixtures can be separated into pure substances
BalasHapusExamples of pure substances are 24 carat gold, distilled water or aquades, and pure iron. Pure substances have certain properties which are always the same, eg pure air always has a melting point or a melting point of 0oC and a boiling point of 100 ° C at atmospheric pressure 1. If an air sample under atmospheric pressure 1 atmosphere does not boil at 100 ° C, we can say it is impure.
HapusExamples of pure substances
(A) carbon,
(B) copper,
(C) gold,
(D) iron,
(E) lead,
(F) of silver,
(G) sulfur,
(H) zinc,
(I) mercury
The mixture contains two or more kinds of constituents. Pure substances always have the same properties, while the mixture may have different properties depending on the composition of its constituent components. This is because each of the compounding substances still possess the properties of origin.
For example, sugar water is a mixture of sugar and air, the sweetness of sugar water may differ depending on the amount of sugar component present in the sugar air. You may have had a drink that is too sweet or too tasty which according to the difference in the amount of sugar added to the drink.
Most of the material we know in everyday life is a mixture. The various types of water we know in everyday life, such as well water, tap water, bottled water, pond water, river water, and sea air are a mixture
What a way to separate the mixture in addition to crystallization and distillation?
BalasHapus1. Separation of Mixture by Decantation
Hapus2. Separation of Substances by Filtration (Filtration)
3. Separation of Substances by Distillation (Distillation)
4. Separation of Substances by Sublimation
5. Separation of Substances by Evaporation
6. Separation of Substances by Chromatography
7. Separation by means of centrifugation (Aspire)
8. Separation with the separation funnel
9. Separation by Crystallization
10. Separation of mixture with Amalgamation
hei weni....!!! What does the element krypton look like?
BalasHapusKrypton is a chemical element with the symbol Kr and atomic number 36. Krypton group of members into 18 and the 4th period. Krypton a noble gas that is colorless, odor, and taste. Krypton occurs in the atmosphere and is separated by air fluid categorization. Krypton is often used with some other gases in incandescent lamps. At low temperatures, the buffer krypton is shaped as liquid or solid. The krypton boiling point is -243.81 ° F (-153.23 ° C), and the freezing point is -251.27 ° F (-157.37 ° C).
HapusKrypton, like other noble gases can be used for illumination and photography. Krypton has a number of spectrum outlines, and high Krypton light in plasma has an important role in many high-power lasers.
Natural krypton is a mixture of six isotopes. Isotopes are atoms that recognize both positive electrical units but have different neutron numbers. The number of positive electrical units (atomic number) can determine the existing elements, while the total number of positive electrical units (atomic number) and neutrons determine the atom's weight. The isotopes of all kryptons have 36 units of positive electricity and are named for atomic weights. Krypton-84, has 48 neutrons, is the most common isotope and constitutes 57% natural krypton.
Physical Properties
Krypton gas is a nadir gas. Krypton is green and the spectral mark is orange. And one of the products of uranium division. The hardened krypton is of a crystal with a centered cube structure, which is the property of all the noble gases. Krypton has an inert (non-reactive) and stable properties, so krypton acts as a shield to protect other unstable materials against air. The concentration of Krypton in the atmosphere is 1 ppm. It can be known from the liquid air by a small distillation. The amount of Krypton in space is uncertain, as does the amount obtained from meteoric activity and from solar storms. Measurements in determining the amount of Krypton are suggested to bestow Krypton inside a chamber.
on your blog about Pure Substances and Mixtures said that most are mixtures, which are combinations of two or more pure substances in variable proportions in which the individual substances retain their identity!!so,how to maintain that identity??
BalasHapusBy maintaining chemical composition and fixed characteristic properties. Oxygen, for example, is a pure chemical which is a colorless and odorless gas at 25 ° C. Very little of the material sample consists of pure substance; Instead, most are mixtures, which are a combination of two or more pure substances in the proportion of the variable in which each of the substances, so that the two mixtures each retain their identity
Hapuschemical change, what does it mean?? Please tell me more about this!
BalasHapusChemical change is a change accompanied by the formation of a new substance. Chemical changes are also known as chemical reactions. The occurrence of chemical changes can be identified with the formation of sediment or gas, as well as the occurrence of discoloration. The formation of gas bubbles is the result of chemical changes. Chemical changes may also lead to sediment formation. Unexpected color changes or odor release also often show chemical changes.
HapusExamples of chemical changes are decaying, burning, cooking, and rusting including from chemical changes as they produce substances that are entirely new chemical compounds. For example, wood burns to ash, carbon dioxide, and water. When exposed to water, iron becomes a mixture of some hydrated iron oxide and hydroxide. Yeast fermentes to produce alcohol from sugar.
For example, the color of the chromium element is determined by the state of the oxidation; A single chromium compound will only change color if it undergoes oxidation or reduction reactions. Heat from cooking eggs changes the interaction and form of protein in egg whites, thus changing the molecular structure and turning the egg whites from translucent to opaque.
Chemical changes have many benefits. Almost all industries that produce raw materials use the principles of chemical change or chemical reactions. In the plastics industry, organic substances derived from natural gas and petroleum are converted through chemical reactions and processes into plastics, eg polyethylene (PE), polypropylene (PP), and polyvinylchloride (PVC).
So what is the conclusion of the material that you post?
BalasHapusHetrogeneous mixtures are mixtures are those in which the components are not evenly distributed over the major component/constitute of the mixture
HapusThe very (unevenly distributed) form of heterogeneous mixtures is the reason why most people shake the mixtures before using them
Ex. Milk of magnesia, copper sulphate
Homogeneous mixuters are those in which the components are evenly distrubuted over the major component/constitute of the mixture
Ex. Blood, milk
How to separate the mixture there are 10 = Separation of Mixture by Decantation, Separation of Substances by Filtration (Filtration), Separation of Substances by Distillation (Distillation), Separation of Substances by Sublimation, Separation of Substances by Evaporation, Separation of Substances by Chromatography, Separation by means of centrifugation (Aspire), Separation with the separation funnel, Separation by Crystallization, Separation of mixture with Amalgamation
A substance that is something that occupies space and has mass.
Solid substances: iron, Nickel, Aluminum, gold, silver
Liquid: water, alcohol, mercury, syrup, soy sauce
Gas substance: oxygen, Nitrogen, helium, ozone, fluorescence
atom and element have similar and different. can you explain similar and different about that?
BalasHapus